In markets where product quality claims are self-reported, third-party laboratory testing serves as the primary mechanism for independent verification. For research peptides, external analysis evaluates whether a compound meets its stated purity, identity, and concentration specifications — without reliance on the supplier’s own testing infrastructure.
This article explains how third-party testing works, what a credible Certificate of Analysis contains, and how to use these documents to compare suppliers with confidence.
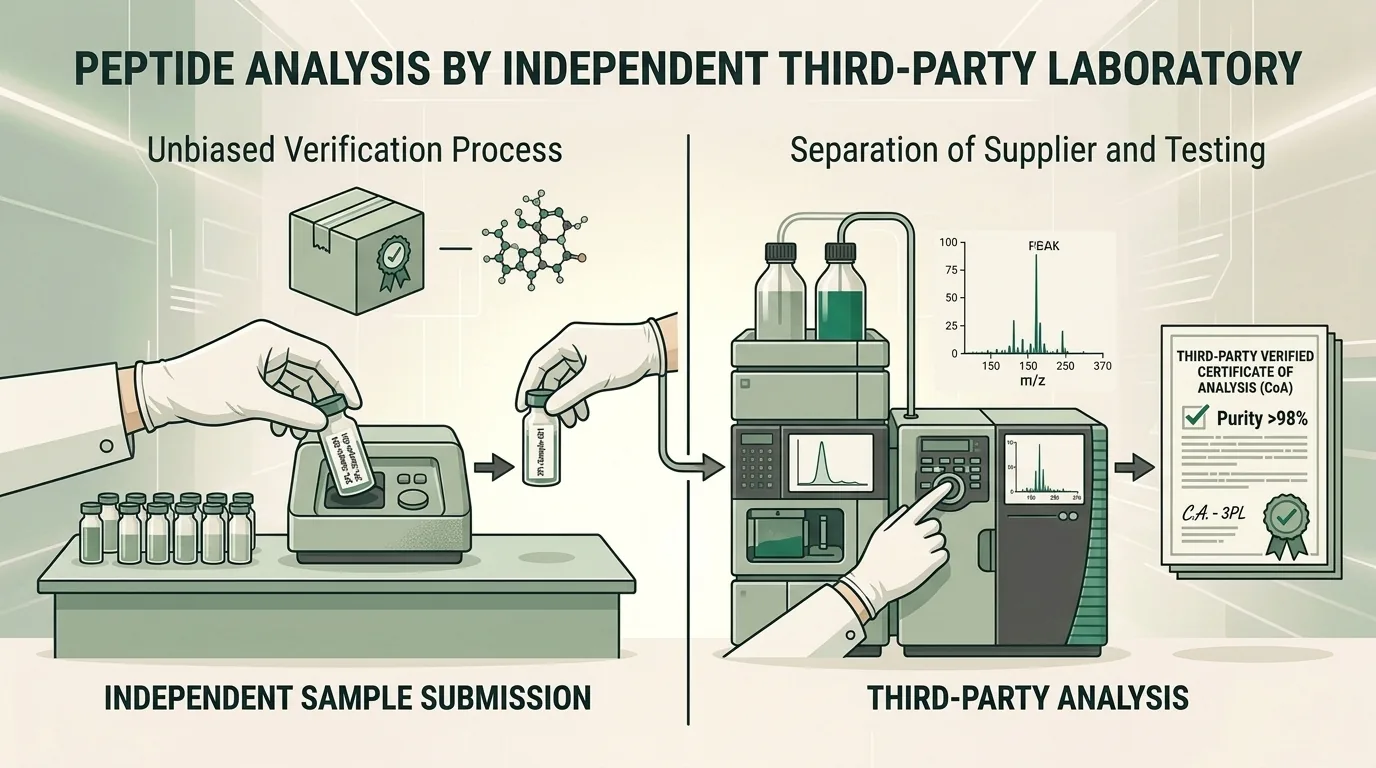
Why Third-Party Testing Matters
When a supplier performs its own quality testing, there is an inherent conflict of interest: the same organization that profits from selling a product is also certifying its quality. Third-party testing eliminates this conflict by introducing an independent laboratory with no financial stake in the results.
Independent verification is particularly critical in the peptide market, where synthesis complexity creates multiple points where purity, identity, or concentration can deviate from specifications.
The Testing Process: Step by Step
Third-party peptide analysis follows a standardized sequence designed to verify both identity and purity:
Step 1: Sample Submission
A representative sample from each manufacturing lot is submitted to the independent laboratory. Proper chain-of-custody documentation ensures the sample’s integrity from supplier to lab.
Step 2: Identity Confirmation via Mass Spectrometry
Mass spectrometry (MS) measures the molecular weight of the compound. By comparing the observed mass-to-charge ratio against the expected molecular weight of the target peptide, the lab confirms the correct amino acid sequence was synthesized. This step answers: Is this the right molecule?
Step 3: Purity Analysis via HPLC
High-Performance Liquid Chromatography separates the sample into its individual components based on chemical properties. The resulting chromatogram shows the proportion of the target peptide relative to impurities, degradation products, and synthesis byproducts. This step answers: How pure is it?
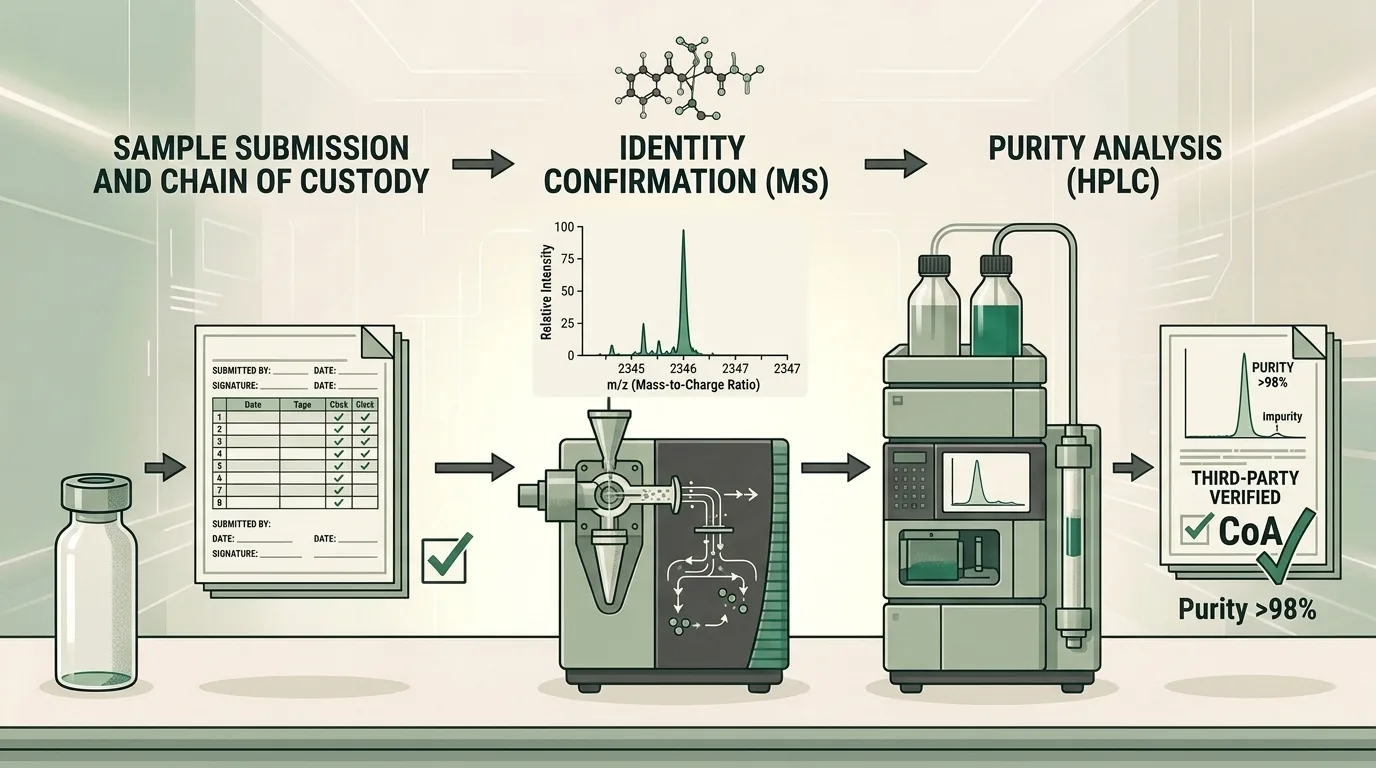
Step 4: COA Generation
The laboratory compiles results into a Certificate of Analysis (COA) — the formal document that summarizes all findings for the tested lot.
Anatomy of a Certificate of Analysis
A complete COA provides all the information needed to evaluate a specific lot’s quality. Here is what each section means:
| COA Field | What It Shows | What to Look For |
|---|---|---|
| Lot / Batch Number | Unique identifier for the manufacturing run | Should be specific — generic or missing lot numbers are a red flag |
| HPLC Purity | Percentage of target compound in sample | 98%+ for research-grade; look for specific numbers, not ranges |
| Molecular Weight (MS) | Observed vs. expected mass of the compound | Should closely match the theoretical molecular weight |
| Testing Laboratory | Name of the facility that performed analysis | Should be a named, identifiable third-party lab |
| Date of Analysis | When testing was performed | Should be recent relative to the product’s availability |
| Appearance | Physical description of the sample | Typically “white to off-white lyophilized powder” for peptides |
Internal Testing vs. Third-Party Testing
Both internal and external testing have roles in quality assurance, but they serve different purposes:
| Factor | Internal Testing | Third-Party Testing |
|---|---|---|
| Independence | No — same entity produces and tests | Yes — independent laboratory |
| Bias potential | Higher — financial incentive to pass | Lower — no stake in results |
| Speed | Faster — on-site equipment | Slower — shipping and lab scheduling |
| Cost to supplier | Lower (existing infrastructure) | Higher (external lab fees) |
| Credibility | Limited without external validation | Higher — independent verification |
| Best used for | In-process quality checks | Final release verification |
How MHS Longevity Approaches Testing
Every MHS Longevity compound undergoes independent third-party analysis before release. Our process includes:
- Lot-specific COAs generated for every manufacturing batch
- HPLC and mass spectrometry performed by independent accredited laboratories
- Results verified against specifications before any product is made available
- COAs available for review upon request or accessible through the product page
Step 1: Request the COA for the specific lot number of your order — not a "sample" or generic COA.
Step 2: Verify the testing laboratory is a named, identifiable facility. Search for the lab independently to confirm it exists.
Step 3: Check that the analytical methods listed (HPLC, MS) are appropriate for peptide analysis. UV-only testing is insufficient.
Step 4: Compare the purity result against the supplier’s stated specification. A legitimate 98%+ result should include the specific decimal (e.g., 98.7%), not a suspiciously round number.

Key Takeaways
Key Takeaways
- Third-party testing eliminates the conflict of interest inherent in self-reported quality claims
- A credible COA includes lot-specific data, named testing laboratory, HPLC purity, and mass spectrometry confirmation
- Independent verification via HPLC and MS is the standard for peptide identity and purity analysis
- Red flags include generic COAs, missing lab identification, and UV-only testing methods
- The combination of internal quality checks and third-party final verification provides the strongest quality assurance framework
Frequently Asked Questions
Every manufacturing lot should be tested independently. A COA from a previous lot does not apply to subsequent production runs, as synthesis conditions and raw materials can vary between batches.
HPLC measures purity by separating sample components. LC-MS (Liquid Chromatography-Mass Spectrometry) combines HPLC separation with mass spectrometry identification, providing both purity and identity data in a single analysis.
In most cases, yes. Reputable suppliers will identify the testing laboratory on the COA, and you can independently verify results by contacting the lab with the lot number.
BPC-157 — Verified 99%+ Purity
Every lot independently verified by third-party HPLC and mass spectrometry analysis. Certificate of Analysis available.


