The term "research-grade" is used widely in the peptide industry, but its meaning is not standardized. Unlike pharmaceutical-grade compounds governed by USP or EP monographs, "research-grade" lacks a single regulatory definition — which means the bar varies significantly from one supplier to the next.
Understanding what this label can and should represent is essential for any researcher evaluating compound quality. This article examines the documentation, testing, and production standards that separate credible research-grade peptides from loosely labeled products.
What Does Research-Grade Actually Mean?
At its core, "research-grade" indicates a compound manufactured to a defined purity standard and accompanied by documentation verifying that standard. Reputable suppliers interpret this as a minimum purity threshold of 98% or higher, verified by independent analytical methods.
However, because no regulatory body enforces a universal definition, some suppliers use the term loosely — applying it to products without third-party verification, incomplete batch records, or inconsistent manufacturing processes.
The Three Pillars of Research-Grade Quality
Credible research-grade designation rests on three interconnected standards: purity verification, batch documentation, and manufacturing consistency.
1. Purity Verification
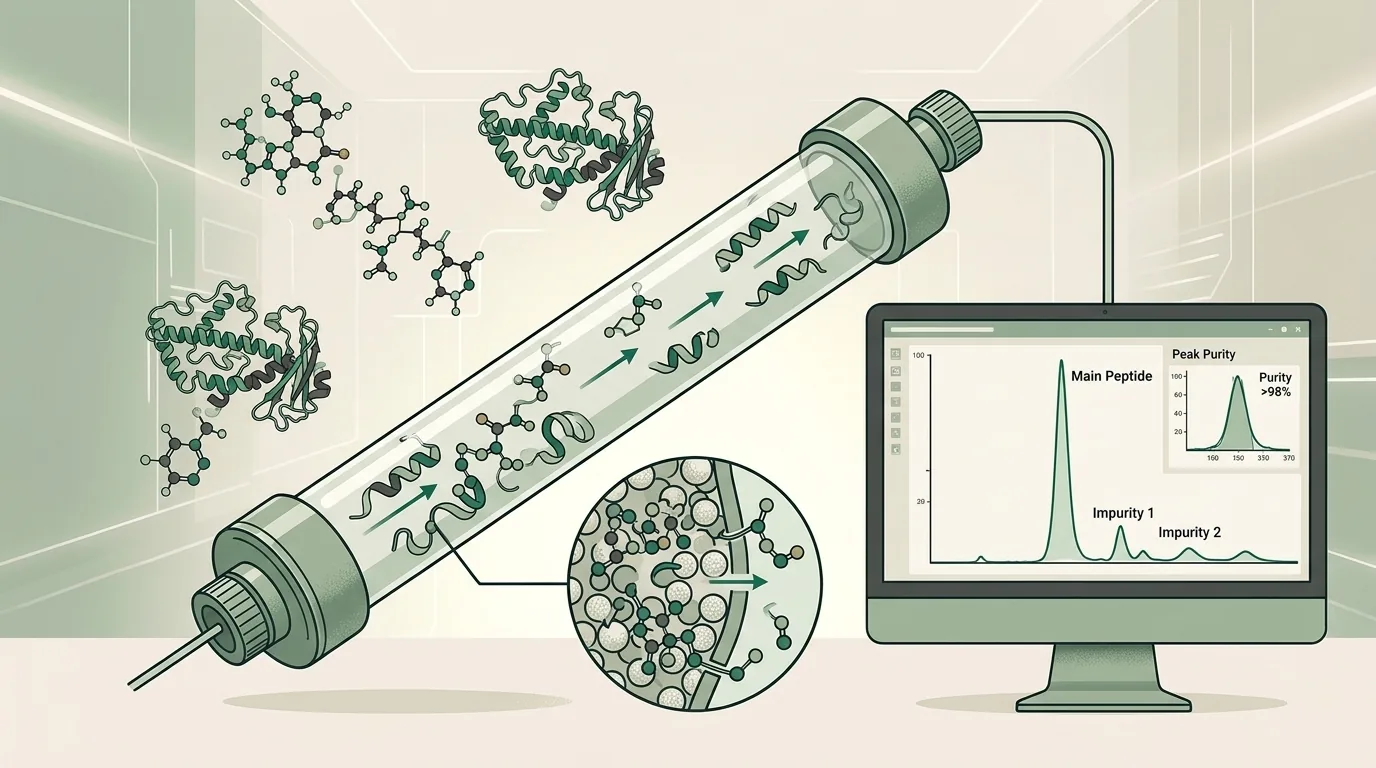
Purity is the primary metric. It represents the percentage of the target compound in a sample, as measured by analytical testing. The two standard methods are:
- HPLC (High-Performance Liquid Chromatography) — Separates sample components to measure the proportion of the target peptide versus impurities, degradation products, and synthesis byproducts.
- Mass Spectrometry (MS) — Confirms molecular identity by measuring the mass-to-charge ratio of the compound, verifying the correct amino acid sequence was synthesized.
Together, HPLC and MS answer two critical questions: Is this the right compound? and How pure is it?
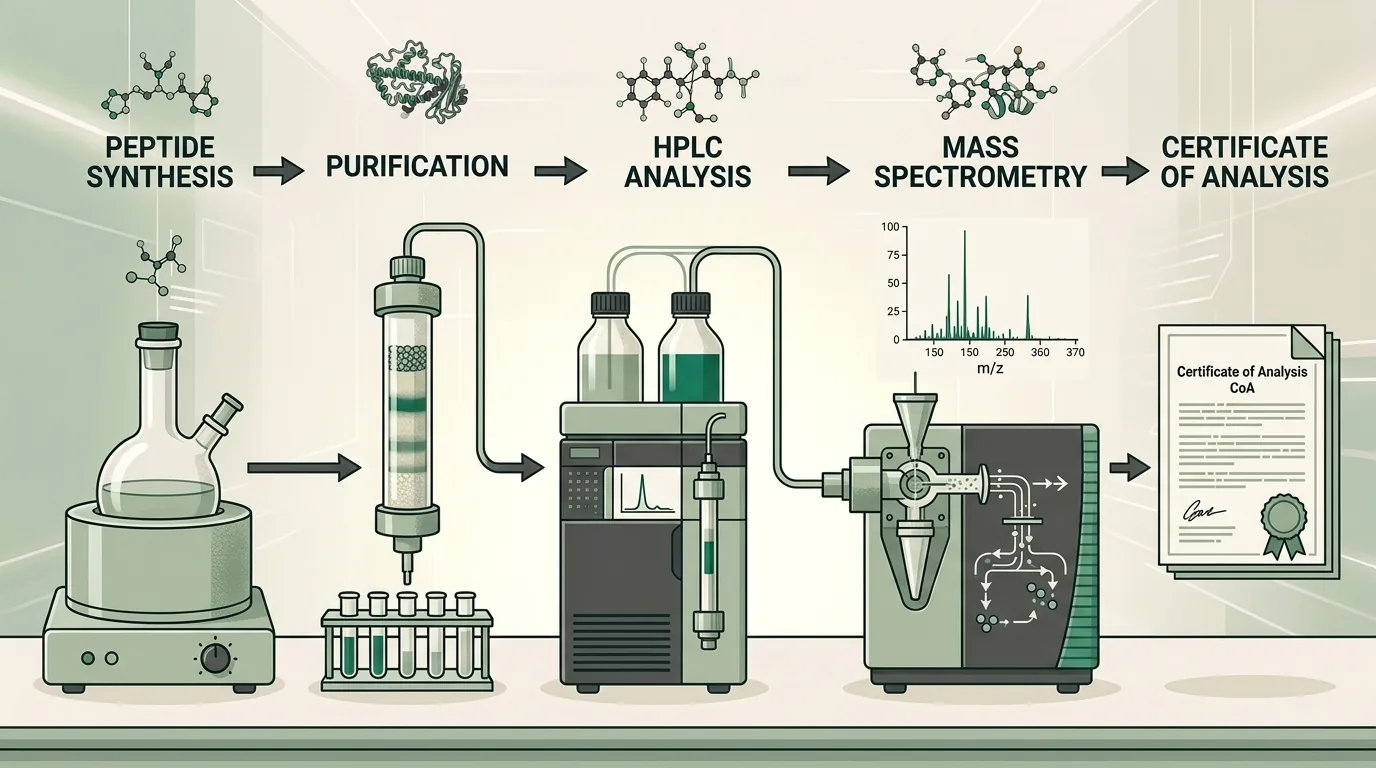
2. Batch Documentation
Every lot of a research-grade compound should be traceable to a specific Certificate of Analysis (COA). A credible COA includes:
- Lot/batch number for traceability
- HPLC purity percentage (typically 98%+ for research-grade)
- Mass spectrometry confirmation of molecular weight
- Testing laboratory identification (ideally third-party)
- Date of analysis
- Physical appearance and form (lyophilized powder, solution, etc.)
3. Manufacturing Consistency
Research-grade quality requires consistent production practices — not just a single good batch. This includes controlled synthesis environments, documented standard operating procedures, and lot-to-lot consistency verification.
Purity Grade Comparison
Not all purity designations are equal. The following table outlines common grade levels encountered in the peptide market:
| Grade | Typical Purity | Verification | Use Case |
|---|---|---|---|
| Cosmetic / General | 70–90% | Internal testing only | Topical formulations |
| Research-Grade | 95–99%+ | HPLC + MS, third-party available | Laboratory research |
| Pharmaceutical-Grade | 99%+ | GMP, USP/EP monographs | Clinical / human use |
How to Evaluate a Supplier’s Research-Grade Claims
When assessing whether a supplier’s “research-grade” label is meaningful, consider the following checklist:
- Request the COA — A credible supplier provides lot-specific COAs on request or with every order. If the COA is unavailable or generic across lots, proceed with caution.
- Verify third-party testing — Look for COAs from independent laboratories rather than in-house testing alone.
- Check the analytical methods — HPLC and mass spectrometry should be standard. UV-only testing is insufficient for peptide purity verification.
- Assess consistency — Ask about manufacturing practices, storage conditions during transit, and whether the supplier maintains documented SOPs.
- Review the purity threshold — Reputable research-grade suppliers target 98%+ purity. Be cautious of vague claims like “high purity” without specific numbers.
UV spectrophotometry measures absorbance at specific wavelengths to estimate concentration, but it cannot distinguish between the target peptide and structurally similar impurities that absorb at the same wavelength. HPLC separates individual components before measurement, providing a much more accurate purity profile.
Mass spectrometry adds a second dimension by confirming the molecular identity of the compound — ensuring the chromatographic peak corresponds to the correct peptide sequence, not a co-eluting impurity.

Key Takeaways
Key Takeaways
- "Research-grade" lacks a universal regulatory definition — standards vary by supplier
- Credible research-grade compounds are verified by HPLC and mass spectrometry, ideally through third-party laboratories
- A complete Certificate of Analysis (COA) with lot-specific data is the minimum documentation standard
- Purity thresholds of 98%+ are the benchmark for reputable research-grade peptides
- Manufacturing consistency and documented SOPs separate reliable suppliers from inconsistent ones
Frequently Asked Questions
No. Research-grade compounds are manufactured and tested for laboratory use, while pharmaceutical-grade compounds meet additional regulatory standards (GMP manufacturing, USP/EP monographs) and are approved for human use. Research peptides are not intended for human consumption.
Reputable suppliers target 98% or higher purity, as measured by HPLC. Some compounds may reach 99%+ depending on synthesis complexity and purification methods.
Look for lot-specific COAs (not generic), third-party laboratory identification, specific analytical methods listed (HPLC, MS), and contact the testing laboratory directly if you have concerns about authenticity.
Explore Our Research-Grade Peptides
Every MHS Longevity compound is verified to 99%+ purity through independent third-party laboratory analysis.
Shop Peptides

